Frequencies other than 64 MHz could cause device damage, excessive heating, and serious patient injury. Do not conduct MRI scans with nonproton scanning frequencies (such as 13C, 23Na, or 31P). If you are unclear what implants are present, perform an X‑ray to determine the implant type and location. 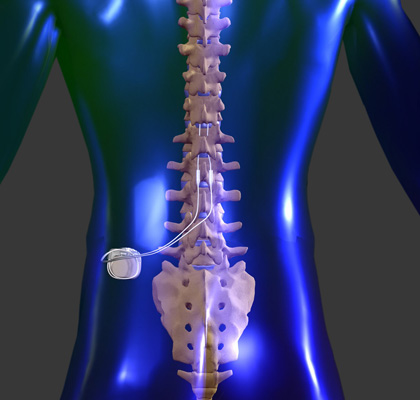 
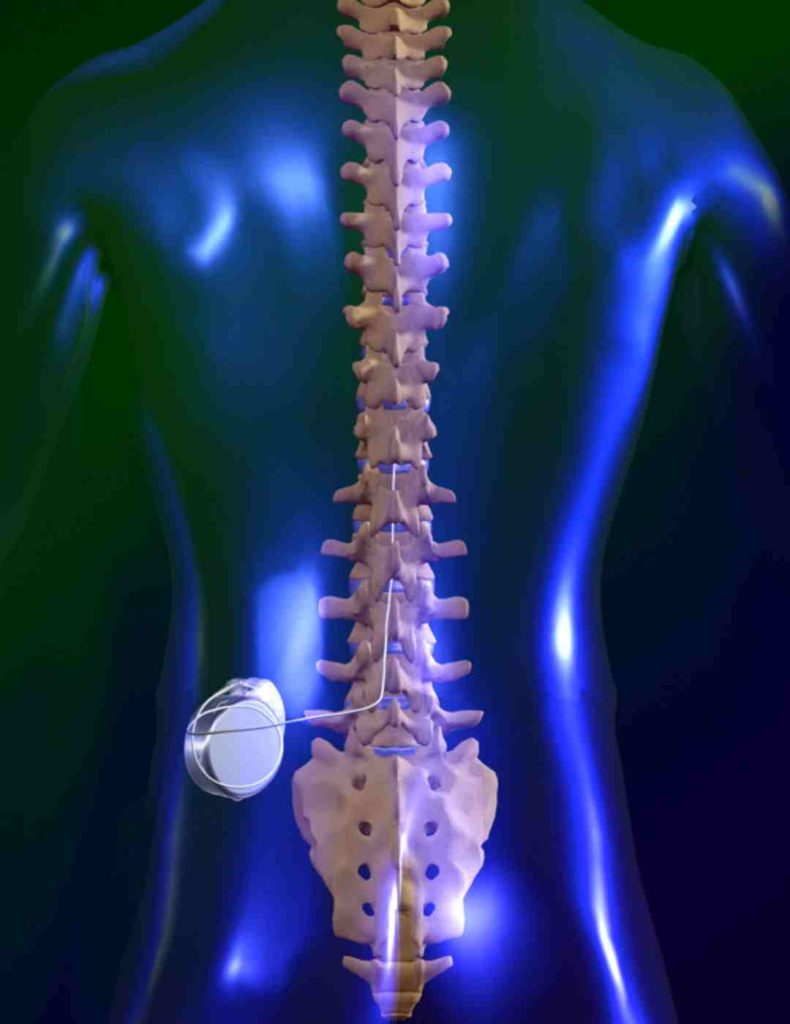
Do not conduct an MRI scan if any conditions or implants prohibit it, as serious patient injury could occur. Of all medical device implants, the most restrictive MRI exposure requirements must be used. Prior to an MRI examination, determine whether the patient has multiple medical device implants, either active medical device implants (such as deep brain stimulation systems, implantable cardiac defibrillators, pacemakers) or passive medical device implants (such as spinal hardware, stents). MRI scans may result in excessive heating of the lead electrodes and serious patient injury. Do not perform an MRI scan on patients who have multiple MR Conditional neurostimulation systems for pain (multiple IPGs for pain). Do not perform an MRI scan on patients who have an external neurostimulation trial system or any components that are not fully implanted. The MRI scan may result in excessive heating of the system and serious patient injury. Do not perform an MRI scan on patients who have any portion of their implanted system exposed due to skin erosion. This can be confirmed with X-ray imaging of the neck, head, and extremity regions or by referring to the patient records. Head or extremity MRI scans can be conducted safely using a Detachable Head or Extremity RF transmit-receive coil when no parts of the implanted neurostimulation system are within the transmit-receive coil according to the conditions specified for each system in the MRI labeling. MRI scans of implants that are not located in approved locations can possibly result in increased unintended stimulation, excessive heating at the lead electrodes, and serious patient injury. Lead tips can be located at different spinal epidural levels. Two leads should travel in close proximity to one another from the IPG to the spine. The MR Conditional leads must be implanted in the epidural space and routed subcutaneously to the IPG pocket. Location of implanted system. To meet the MR Conditional requirements, components must be implanted according to the approved locations specified by the MRI labeling. MRI scans of nonfunctional leads may result in excessive heating occurring at the location of the implanted lead electrodes and serious patient injury. There may be a problem with the implanted lead(s)” message displays when attempting to enter MRI mode on the patient controller. Do not perform an MRI scan on patients when the “MRI is Not Advised. Do not perform an MRI scan on patients who have an incomplete neurostimulation system, where a lead is present without the IPG or disconnected from the system. Serious patient injury could occur.Ībandoned devices. Do not perform an MRI scan on patients who have any components of a neurostimulation system that are unapproved for use in an MR environment. The following warnings, precautions, and potential adverse events apply when performing MRI scans on Abbott MR Conditional SCS/DRG systems. Integrated whole-body transmit coil: SAR ≤ 0.1 W/kgĭetachable head transmit-receive coil: Normal operating modeĭetachable extremity transmit-receive coil: Normal operating modeģ0 minutes total of active scan time per session Head scans: Integrated whole-body transmit coil with any receive coil OR detachable head transmit-receive coilĮxtremity scans (including hip and shoulder): Integrated whole-body transmit coil with any receive coilĮxtremities (all except hip and shoulder): Detachable extremity transmit-receive coil Any body part scans: Integrated whole-body transmit coil with any receive coil
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
